Topics in Photographic Preservation 2007, Volume 12, Article 11 (pp. 55-63)
Presented at the 2007 Joint PMG/ICOM-CC WGPM Meeting, Rochester, New York
The article concerns studies of micromycetes carried out in several old photographic collections (materials dating from 1839 - 1912). Most of the species revealed during research belong to anamorphic fungi. Results of the research proved that the destruction of photographic images does not depend on characteristics of photographic layer and substrate of the basis. Laboratory trials with membrane cellulose filters showed that 86% of revealed fungi are capable to destroy this substrate. Therefore they can be considered potential biodestruction agents of photo images.
St.Petersburg is a city world known for its rich cultural heritage. The terms of storage and usage of historical valuables can make up to hundreds of years. Destruction of architectural monuments and artworks, items stored in museums, libraries and archives is brought about by chemical and physical factors speeding up the ageing of materials, as well as by the activity of various microorganisms. Various microorganisms settling on archival materials and artworks include microscopic fungi, actinomycetes, bacteria, algae. They can be transported by air flow when spores, conidia, fragments of fungal hyphae travel with dust particles and other pollutants.
In a humid environment, these propagules easily germinate and develop, further forming biodestructive complexes of microorganisms of a wide range. For museum storages with controlled temperature and humidity levels, it is vital to avoid dramatic temperature fluctuations that can be caused, for example, by heating turn-offs in winter time, a situation quite characteristic for St.Petersburg. Dramatic lowering of temperature leads to growth of relative humidity in museum and archive storages and in some cases to moisture condensation. In the spaces where good air circulation is provided, the situation quickly comes back to normal. However, in the cabinets where photographic materials are stored such situation can easily instigate growth of fungi on objects.
Microscopic fungi take priority among various groups of organisms developing within the conditions of museums and archives. The ever declining ecological situation in urban environments leads to dramatic growth of micromycetes and their participation in destructive processes, as well as to the increase in number of mycotic diseases, especially among museum employees.
A separate problem is the massive growth of mould in storages and archives which, besides causing destruction of museum items, can be dangerous for the health of the staff. The last decades witnessed more frequent cases of diseases caused by human opportunistic pathogens.
This should evoke a special concern to those kinds of fungi that potentially can become human opportunistic pathogens (de Hoog, 1993).
Pathogenicity of micromycetes depends on a complex of adaptive features allowing specific micromycetes to colonize and invade the host organism. Such adaptive qualities as ability to survive at temperatures up to 37°C and even over, ability for extra cellular secretion of aspartic proteinases and phospholipases, among others, are traditionally referred to as pathogenicity factors. The intensity level of these characteristics defines the pathogenicity level of the fungus.
Phospholipases and proteinases are important factors of the pathogen's invasion into the host organism tissues, destruction of cellular membranes and surface molecules as well as destruction of immune system cells and molecules (Smolenski et al., 1997; Hube, 1998; Fekete-Forgacs et al., 2000; Ghannoum, 2000; Niewerth, Korting 2001, Shen et al., 2004; Schaller et al. 2005; Fotedar, Al-Hedaithy, 2005).
Mycological research was carried out in the following collections:
Salted Paper Prints, 1839, by William Henry Fox Talbot (St.Petersburg Branch of the Archive of Russian Academy of Sciences; curator O.V. Iodko, director IV. Tunkina) - 6 pages.
Dyagilev Album, 1870–1890 (Institute of Russian Literature of the Russian Academy of Sciences; curator M.V. Radyukova, Director N.N. Skatov). Albumen photographs on album pages - 12 pages.
Russian Turkestan, 1895, by Paul Nadar. (Material Culture Institute of the Russian Academy of Sciences, curator - G.V. Dluzhnevskaya, director E.N. Nosov). Albumen photographs - 15 pages.
Views of Caucasus, 1910. (National Centre of Photography of the Russian Federation; curator E.A. Glushkova, director Z.M. Kolovsky). Albumen photographs in passepartouts - 16 pages.
Views of St.Petersburg, 1912, by Lawrence. (National Centre of Photography of the Russian Federation; curator E.A. Glushkova, director Z.M. Kolovsky). Albumen photographs- 12 pages.
Samples were selected in winter and spring time of 2005. The examination revealed the species composition of micromycetes dwelling on the surface of photographs and in the cellulose base. 51 pages were examined in total, each page containing 1 to 3 images.
The fungi were extracted by direct print method and dry needle method (Methods of experimental mycology, 1982). The cultures were grown using humid camera method and on the following culture media: Czapek, Czapek-Dox, malt agar, yeast extract agar, cellulose substrate. Samples were incubated in Petri dishes at a temperature of 250°C. Starting on the 7th day, the dishes were examined and micromycetes colonies were isolated into pure culture. Incubation period lasted for 21 days. Upon pure cultures isolation, Czapek agar was used for their identification and further storage. Species identification was made using the following identification keys: Barron, 1968; Raper et al., 1968; Ellis, 1971, 1976; Ramirez, 1982; Domsh et al., 1993.
Frequency of occurrence index was defined (separately for each collection) as proportion of samples where fungi was found to the total amount of samples examined. The abundance of fungal species was calculated as percentage of one species' propagules within the total amount of samples received from one collection.
In order to test protease activity of micromycetes, culture medium containing bovine serum albumen was used (Fotedar, Al-Hedaithy, 2005). Upon inoculation, micromycetes were incubated in Petri dishes for 10 days at room temperature. After that, diameters of colony and of clarification area surrounding it were measured. The clarification area forms due to albumen hydrolysis by extracellular protease of the fungus (if such protease exists). Protease activity coefficient was calculated by the following formula:
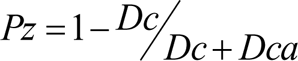
Where Pz is protease activity index, Dc is diameter of colony, Dca is diameter of clarification area.
For the experimental testing of common phospolipase activity of micromycetes (without identification of phospholipase types), egg yolk containing substrate was used (Fotedar, Al-Hedaithy, 2005). Upon inoculation, samples were incubated in Petri dishes at room temperature for 10 days. After that, diameters of colony and of precipitation area surrounding it were measured. Precipitation area is formed as a result of chemical reaction of calcium salt with egg yolk phospholipids caused by the extra-cellular fungus phospholipase. Phospholipase activity index is calculated at the same formula as the one used for protease activity replacing the measurement of clarification area with that of precipitation area.
Experiment testing growth and survival of fungal cultures at 37°C was held on oat agar. The cultures were incubated for 7 days at a temperature of 37°C. After that, amount of growth (if such happened) was measured, and check inoculation was made into a new agar plate at a room temperature, in order to check the fungi's viability. The cultures that survived and showed limited growth, were estimated as those capable of tolerating the temperature of 37°C.
Biodestructive activity of isolates was evaluated by their ability for growth on liquid mineral substrate with cellulose being the only source of organic matter. The parameters measured were the growth rate of culture and weight loss of cellulose. The experiment was held for 30 days at a temperature of 250°C. During the experiment, antibiotics (penicillin, erythromycin) at 100mg/100ml concentration were used to suppress possible bacterial contamination.
The research made resulted in detection of 23 fungi species belonging to 11 genera from: Deuteromycotina (anamorphic fungi) and Ascomycotina (ascomycetes) subdivisions, as well as sterile white and dark mycelia which seems difficult to define a species identity for at the moment. The largest species diversity was found for Penicillium: 10 species, whereas Aspergillus, Chrysosporium and Oidiodendron were represented with 2 species each and the remaining genera with just one species each.
The largest diversity of species was found on the Views of Caucasus collection (18). Salted Paper Prints and Dyagilev Album collections showed 4 different species each, Views of St.Petersburg and Russian Turkestan collections - 5 species each.
The Salted Prints Collection was 100 per cent affected (chart 1), with dominating isolates being Paecilomyces variotii and singular presence of other species' isolates. Dyagilev Album photographs were affected to a lesser extent (33%).
Chart 1 Distribution of micromycetes found among the collections.
| Collection* | Number of species | Number of isolates | Proportion of photographs affected |
| Salted Paper Prints | 4 | 48 | 100% |
| Dyagilev Album | 4 | 4 | 33% |
| Views of Caucasus | 18 | 50 | 75% |
| Russian Turkestan | 5 | 8 | 33% |
| Views of St.Petersburg | 5 | 10 | 33% |
| Total | 23 | 120 |
* — Collection descriptions are included in the text
In Views of Caucasus collection, affected photographs made 75% of the collection, however the diversity of species was the largest in this collection. So was also the number of isolates (50). Views of St.Petersburg and Russian Turkestan collection were affected to a relatively low extent, 33%, and the number of isolates detected was also relatively small - 10 and 8, correspondingly.
Cladosporium cladosporioides was detected among fungal isolates in every collection. Alternaria alternata isolates were detected in four collections, excluding Russian Turkestan one. Penicillium canescens and P. cyclopium were detected among isolates in 3 collections, whereas Chaetomium sp. - in two of the collections. Paecilomyces variotii prevailing among isolates on salted prints collection was singularly present in Russian Turkestan collection but absent among the variety of fungi on other collections. Other fungi isolates were only found once in each collection.
As has already been mentioned, Salted Paper Prints collection was affected primarily by Paecilomyces variotii micromycetes. In addition, beside Cladosporium cladosporioides and Alternaria alternata, Ulocladium sp. was also detected.
The range of fungi detected in Dyagilev Album collection includes isolates of Cladosporium cladosporioides and Alternaria alternata, as well as Oidiodendron citrinum and Penicillium citrinum.
The largest diversity of fungi species was observed in the Views of Caucases collection. Here, Penicillium prevails with number of species (9), and beside it Aspergillus restrictus, Chrysosporium merdarium and Ch. Pannorum were detected. There is a considerable proportion of dark-colored micromycetes among isolates from this collection. These include 5 species: Alternaria alternata, Cladosporium cladosporioides, Doratomyces microsporus, Oidiodendron sp. and Torula herbarum.
Furthermore, Chaetomium sp. isolates showed a high frequency. This species is known for its high cellulose destructive ability which was proved in our experiments on isolates of this fungus.
In Views of St.Petersburg collection, the following micromycetes were found: Alternaria alternata, Aspergillus versicolor, Cladosporium cladosporioides, Penicillium canescens and P. cyclopium
In Russian Turkestan collection, Chaetomium sp., Cladosporium cladosporioides, Paecilomyces variotii, Penicillium canescens and P. cyclopium were detected.
For the comparison of micromycetes detected in different photographic collections, the index of dynamic structure of fungi complexes was used (Chart 2). First the amount of micromycetes belonging to specific class of growth rate (Kr) was evaluated. The difference in growth rate between classes was accepted as 0.05 mm/hour (Polyanskaya et al., 1988). Comparing the indexes of slow growing (belonging to 1–2 class with radial growth rate up to 0.10 mm/hour) and fast growing micromycetes (belonging to 3–7 Kr classes with growth rate from 0.11 to 0.61 mm/hour) leads to a conclusion that the part of fast growing species in Salted Paper Prints collection is somewhat larger than the corresponding indexes in albumen photograph collections.
Chart 2 Structural characteristics of micromycetes complexes in the examined photographic collections.
| Collection | Micromycetes species | Frequency (%) | Abundance of species (%) | Growth rate (Kr classes) |
| Salted Paper | Alternaria alternata | 17 | 2.1 | 5 |
| Prints | Cladosporium cladosporioides | 17 | 2.1 | 2 |
| Paecilomyces variotii | 100 | 93.7 | 6 | |
| Ulocladium sp. | 17 | 2.1 | 4 | |
| Dyagilev | Alternaria alternata | 8 | 25 | 5 |
| Album | Cladosporium cladosporioides | 8 | 25 | 2 |
| Oidiodendron citrinum | 8 | 25 | 2 | |
| Penicillium citrinum | 8 | 25 | 2 | |
| Russian | Chaetomium sp. | 19 | 37.5 | 6 |
| Turkestan | Cladosporium cladosporioides | 6 | 12.5 | 2 |
| Paecilomyces variotii | 6 | 12.5 | 6 | |
| Penicillium canescens | 6 | 12.5 | 3 | |
| P. cyclopium | 13 | 25 | 6 | |
| Views of Caucasus | Alternaria alternata | 6 | 2 | 5 |
| Aspergillus restrictus | 6 | 2 | 3 | |
| Chaetomium sp. | 25 | 10 | 7 | |
| Chrysosporium merdarium | 6 | 2 | 2 | |
| Ch. Pannorum | 6 | 2 | 3 | |
| Cladosporium cladosporioides | 19 | 10 | 2 | |
| Doratomyces microsporus | 13 | 4 | 2 | |
| Oidiodendron sp. | 6 | 2 | 2 | |
| Penicillium brevi-compactum | 13 | 8 | 2 | |
| P. canescens | 6 | 2 | 3 | |
| P. chrysogenum | 19 | 6 | 4 | |
| P. cyclopium | 31 | 10 | 6 | |
| P. decumbens | 6 | 2 | 2 | |
| P. dierckxii | 13 | 8 | 2 | |
| P. expansum | 6 | 2 | 4 | |
| P. notatum | 6 | 8 | 3 | |
| P. waksmanii | 19 | 10 | 1 | |
| Torula herbarum | 6 | 6 | 2 | |
| Views of St.Petersburg | Alternaria alternata | 8 | 20 | 5 |
| Aspergillus versicolor | 8 | 10 | 2 | |
| Cladosporium cladosporioides | 8 | 20 | 2 | |
| Penicillium canescens | 16 | 20 | 3 | |
| P. cyclopium | 16 | 30 | 6 |
Micromycetes extracted in Dyagilev Album and Views of Caucasus collections, were tested for cellulose destruction ability. It was discovered that only 3 species (Chrysosporium merdarium, Ch pannorum H Penicillium decumbens, comprising 14% of the total amount of species extracted) are practically unable to develop on cellulose in case of the latter being the only source of organic matter and most likely, these are contaminant fungi brought in by chance. Isolates of remaining species (86%) are able to develop on cellulose and decompose it, to a different extent and at different speed and with favorable conditions given, which leads to the loss in weight and destruction of the material (Fig. 1).
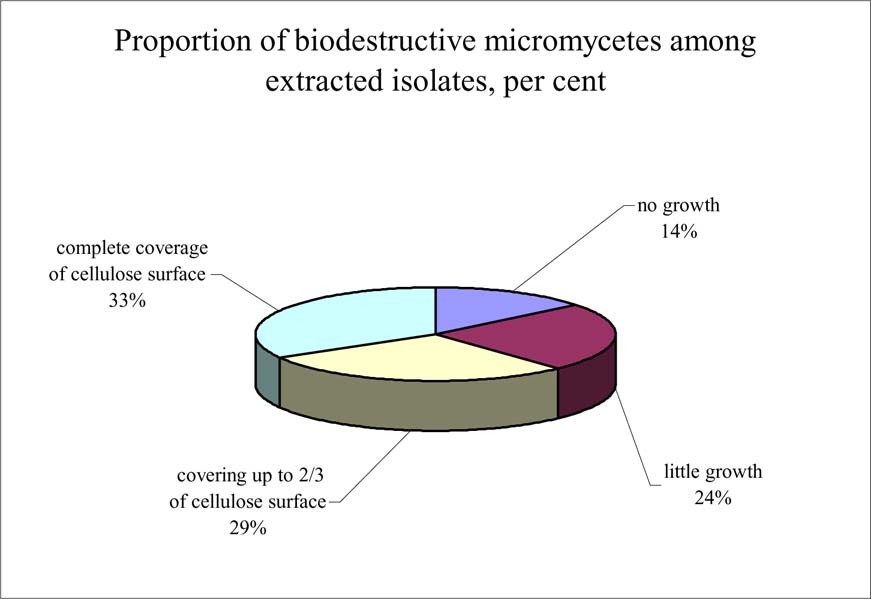
The dry weight loss after the development of Alternaria alternata, Chaetomium sp., Cladosporium cladosporioides, Doratomyces microsporus, P. notatum and Torula herbarum micromycetes on cellulose filters during one month was up to 20% in some cases. The data obtained showed that cellulosolytic ability of microfungi certainly affects the safety of photographs in the conditions favourable for the development of this biodestructors.
Trials measuring protease and phospholipase activity have proved such activity being characteristic for Cladosporium cladosporioides isolate. In the course of experiment, it was discovered that enzyme activity (protease and phospholipase activity) levels of the strain change little in time. The average phospholipase activity value of C. cladosporioides strain equaled 0.089, with the maximum at 0.119 and minimum at 0.069. Average protease activity value of C. cladosporioides equaled 0.172, with maximum at 0.307 and minimum at 0.053. Protease activity gradually decreased during the period of colony growth.
The ability of this strain to grow at a temperature of 37°C has not been proved.
The data received proves that salted paper prints and albumen photographs are prone to being attacked by microscopic fungi irrespective of the difference in physical and chemical characteristics of these substrates. The expanding of research on photographs with various emulsion and base types would allow identifying certain regularities in the formation of fungi complexes that act as biological destructors on archival photographs. Furthermore, it would enable to define further research on the preservation of existing collections and prevention of their further destruction.
The research described was also the first time to examine virulence factors of one of the isolated strains of opportunistic micromycetes. This allows one to conclude that mycotic biota dwelling on photographs contains separate propagules of micromycetes dangerous for human health.
E.V. Bogomolova, A.M. Ivanova, I.Y. Kirtsideli, V.A. Mel'nik*, D.V.Sokolenko**
* Komarov Botanical Institute, Russian Academy of Sciences. St. Petersburg, Russia
** National Centre of Photography of the Russian Federation. St. Petersburg, Russia
The authors are grateful to the curators of photographic collections mentioned for their cooperation. Separate thanks should be expressed to Zakhar Kolovsky, director of the National Centre of Photography of the Russian Federation. His administrative support in the solution of financial questions made a great impulse to the whole research work.
- Methods of experimental mycology / Ed. V.I. Bilay. Kiev. 1982. 346 p. (in Russian).
- Polyanskaya L.M., Triger E.G., Kozhevin P.A., Zvyagintsev D.G. Kinetic description of soil actinomicromycetes complexes structure Microbiologiya, 1988. V. 57(5). P. 854–859.
- Barron G. L. The genera of Hyphomycetes from soil Baltimore. 1968. 364 p.
- Domsh K. B., Gams W., Andersen T.-H. Compendium of soil fungi. London : Acad. Press. 1993. Vol. 1. 859 p.
- Ellis M. B. Dematiaceous hyphomycetes. Kew. Surrey. 1971. 608 p.
- Ellis M. B. More dematiaceous hyphomycetes. Kew. Surrey. 1976. 621 p.
- Fekete-Forgacs Gyure L., Lenkey B. Changes of virulence factors accompanying phenomenon of induced fluconazole resistance. in Candida albicans Mycoses. 2000. Vol. 43. P. 273–279.
- Fotedar R., Al-Hedaithy S.S.A. Comparison of phospholipase and proteinase activity. in Candida albicans and Candida dubliniensis Mycoses. 2005. Vol. 48. P. 62–67.
- Ghannoum M.A. Potential role of phospholipases in virulence and fungal pathogenesis Clinical Microbiology Reviews. 2000. Vol. 13. No. 1. P. 122–143.
- Hoog G.S. de Evolution of black yeasts: possible adaptation to the human host Antonie van Leeuwenhoek. 1993. Vol. 64. P. 321–329.
- Hube B. Possible role of secreted proteinases in Candida albicans infections Rev. Iberoam. Micol. 1998. Vol. 15. P. 65–68.
- Niewerth M., Korting H.G. Candida albicans and the principle of opportunism. An essay//Mycoses. 2002. Vol. 45. P. 253–258.
- Ramirez C. Manual and atlas of the Penicillia. Amsterdam. New York. 1982. 874 p.
- Raper B., Thom C., Fennell D. I. A manual of Penicillia. New York, London, 1968. 875 p.
- Schaller M. Borelli C., Korting H.C., Hube B Hydrolytic enzymes as virulence factors of Candida albicans / Mycoses. 2005. Vol. 48. P. 365–377.
- Shen D.-K., Noodeh A.D., Kazemi A., Grillot R., Robson G., Brugere J.-F. Characterisation and expression of phospholipases B from the opportunistic fungus. Aspergillus fumigatus FEMS Microbiology Letters. 2004. Vol. 239. P. 87–93.
- Smolenski G., Sullivan P.A., Cutfield S.M. Analysis of secreted aspartic proteinases from Candida albicans: purification and characterization of individual Sap1, sap2 and Sap3 isoenzymes Microbiology. 1997. Vol. 143. P. 349–356.
Papers presented in Topics in Photographic Preservation, Volume Twelve have not undergone a formal process of peer review.